Foundry

Arun Ramakrishnan
CTO and Co-Founder, LogicFlo AI

The use of generative AI in medical content creation is accelerating at a pace few could have predicted. For Medical Affairs teams, particularly those in medical communications and medical information, the potential is enormous: faster drafting, streamlined reviews, and near real-time updates to support HCP engagement. But alongside this promise comes a critical reminder—FDA standards have not changed.
No matter how advanced the tool, the responsibility for accuracy, balance, and compliance rests with the company. AI can make it easier to scale high-quality content, but it can just as easily amplify mistakes.
Why This Matters Now: Speed, Scale, and Risk
Large language models (LLMs) and AI-driven drafting tools can shrink timelines from weeks to hours. That efficiency can be game-changing, but it also magnifies risk. An unchecked AI draft that strays from the product label, omits safety information, or implies an unapproved use could trigger regulatory action just as quickly.
The FDA is clear: the same standards apply whether content is written by a human or generated by AI. Companies remain fully accountable for every word distributed. This makes it essential for Medical Affairs teams to rethink workflows so that compliance checks scale with AI output.
The Core FDA Guidances That Matter
Several FDA guidances define the regulatory perimeter for medical and promotional content. For Medical Affairs leaders, these become the framework that AI-enabled systems must follow.
On-Label Consistency
All promotional communications must stay within the scope of the approved indication, dosing, patient population, and conditions of use. Content must be truthful, balanced, and supported by evidence. (REF)Off-Label Scientific Exchange
Companies may share scientific information on unapproved uses, but only under strict conditions. Such communications must be factual, balanced, and accompanied by clear disclosure that the use is not FDA-approved. Importantly, these exchanges must remain scientific, not promotional. (REF)Risk/Benefit Balance
Any communication must present risks with equal prominence and readability as benefits. Omitting material safety facts is considered misleading and remains a frequent cause of FDA enforcement. (REF)Unsolicited Requests
AI used in medical information must only respond to genuine unsolicited requests from HCPs. These responses must be scientific in tone, tailored to the specific question, and balanced with evidence and limitations. (REF)Misinformation Correction
Companies may correct misinformation found in third-party spaces. Corrections must be factual, not promotional, and should use consistent, pre-approved statements (REF). AI may help monitor and detect misinformation, but the corrective response must remain strictly compliant.
Key Risk: Misinterpreting “scientific exchange” as permission for broad off-label messaging. Even with the FDA’s January 2025 clarification, proactive off-label communications carry a high bar. They must be complete, balanced, and free of promotional tone. AI can draft these, but human reviewers must ensure disclosures are included and the communication remains unbranded and non-promotional. (REF)
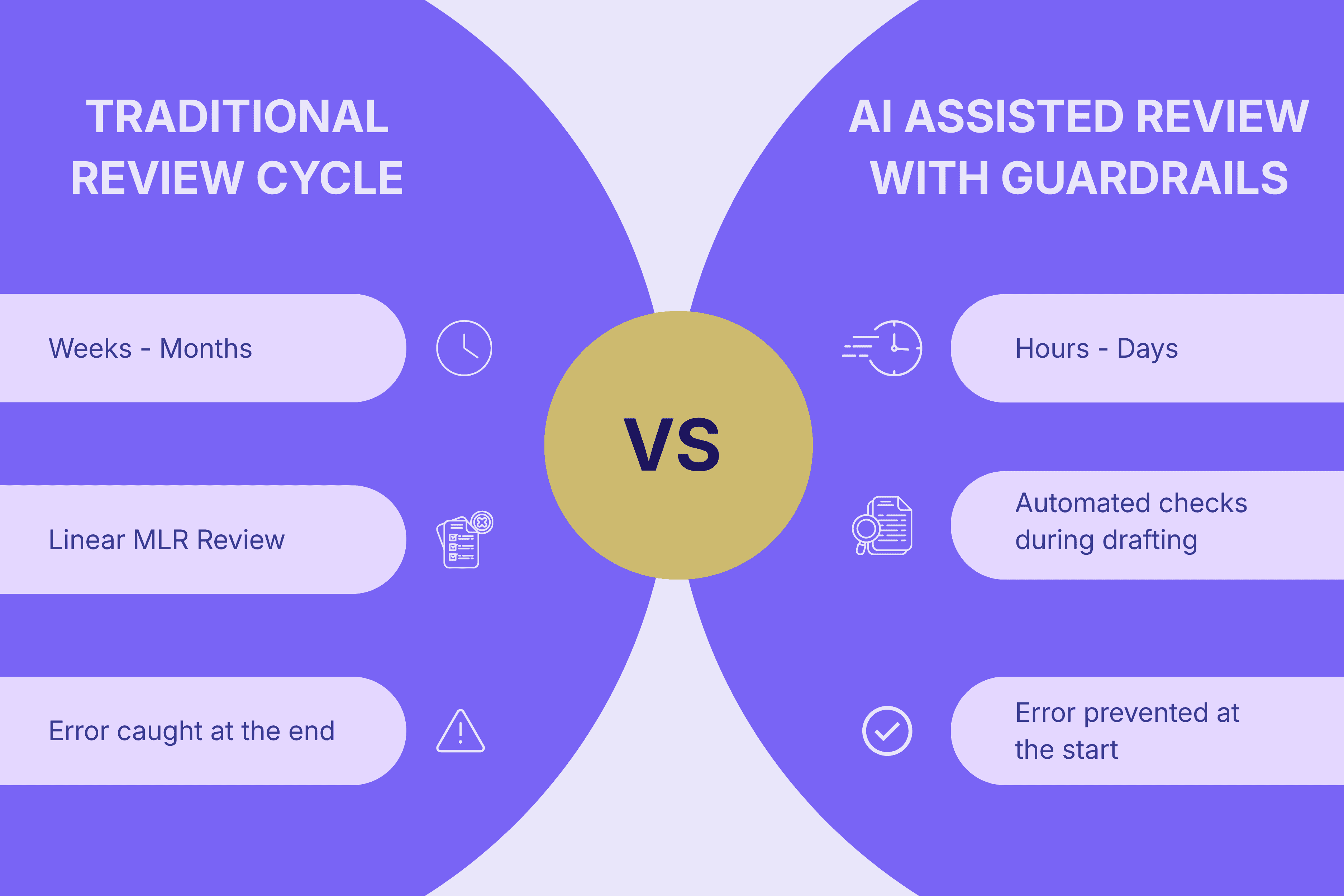
Then vs. Now: Traditional vs. AI-Accelerated Cycles
Medical content development has traditionally been a slow, manual process. Literature reviews, first drafts, and multiple rounds of reference checks could stretch timelines for weeks. AI can compress that timeline to hours, enabling rapid updates and personalized variants.
The challenge is that compliance systems designed for monthly updates now face the pressure of daily or on-demand content generation. This mismatch exposes new operational gaps:
On-Label Drift – Without access to the approved label, LLMs may generate claims outside the permitted scope.
Omitted Safety Information – AI models often highlight benefits without automatically including risks, a major FDA compliance trigger.
Failure to Update with Label Changes – If AI systems are not synced with the latest approved labeling, outdated information can be propagated at scale, effectively misbranding the product.
Feature | Traditional Review Cycle | AI-Assisted Review with Guardrails |
Pace | Weeks to months | Hours to days |
Process | Linear MLR review | Automated checks during drafting |
Risk Management | Errors caught at the end | Errors prevented at the start |
LogicFlo AI’s Role: Embedding Guardrails into the Workflow
To keep pace, Medical Affairs teams need AI that does more than draft. It must actively enforce compliance guardrails. LogicFlo AI is designed with this operational need in mind, serving as a layer of compliance intelligence throughout the content lifecycle.
Trained on FDA Guidance and Product Labeling
Models are anchored to the official label, ensuring they draw from the approved source of truth.Flagging Scope Drift
AI checks claims against the approved indication, dosing, and conditions of use, flagging deviations before they reach review.Enforcing Safety Inclusion
Templates and prompts ensure risk information is always included, with the correct safety statements drawn directly from the label.Cross-Checking During MLR Review
AI assists reviewers by verifying references, spotting missing disclosures, and flagging promotional tone. Tools like Veeva’s MLR Bot are early examples of this approach.Lifecycle Management
AI can scan a company’s full content repository after a label change, flagging outdated materials and prompting timely updates.
Takeaways for Commercial & Medical Affairs
A 2025 report published in August by McKinsey suggests that GenAI will not rewrite the FDA’s rules, but it will reshape how teams meet them (REF). The choice is between reactive compliance - trying to catch errors after the fact, or proactive compliance - embedding guardrails from the start.
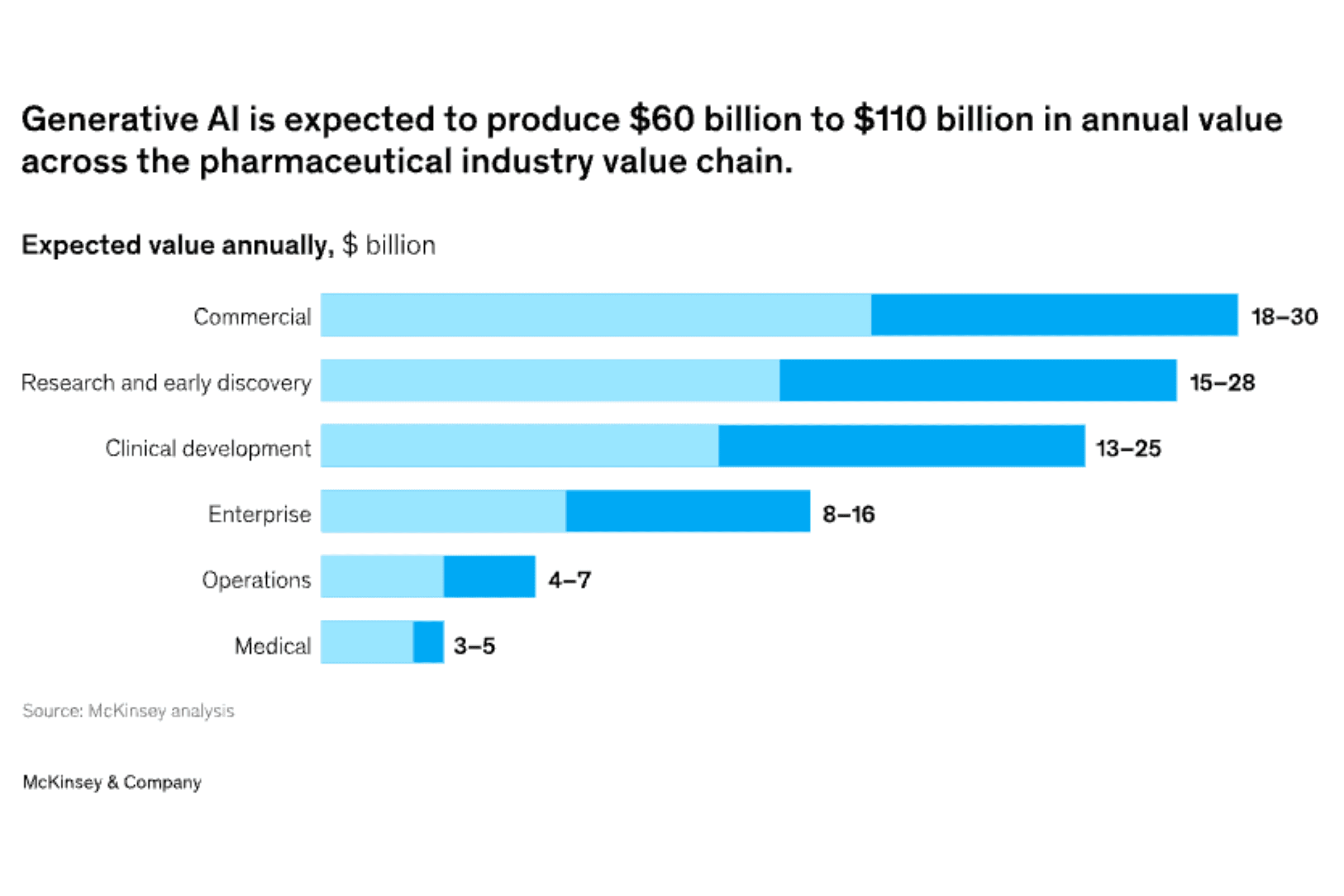
Source: McKinsey Analysis 2024
Pharma leaders should:
Anchor AI systems to FDA guidance and product labeling.
Build workflows that force inclusion of risk information.
Maintain human oversight, especially for scientific exchange.
Treat lifecycle updates as continuous, not episodic.
A recent ZS report suggested 93% of life sciences leaders anticipate increased investments in data, digital, and AI in 2025 (REF). AI can be a powerful accelerant for compliant, high-quality content. But without the right safeguards, it can be just as powerful a liability. The path forward is clear: compliance must remain the foundation, and AI must be built to reinforce it.
View All

Featured In
Why Life Sciences Needs Its Own Integrated Development Environment
Fragmented medical workflows limit AI. Vertical IDEs unify context, improve traceability, reduce risk, and enable reliable agent-driven content creation.

Arun Ramakrishnan
•

Featured In
Bridging The AI Divide: What It Really Takes To Get AI Agents Into Production
95% of AI pilots fail; success requires agents with context, tool integration, guardrails, usable interfaces, and focus beyond prototypes.

Udith Vaidyanathan
•

Featured In
When AI Meets Life Sciences: Separating Hype From Reality
AI turns chaotic data into insight at scale, but human judgment, integration challenges, and accuracy constraints define success.

Udith Viadyanathan
•






